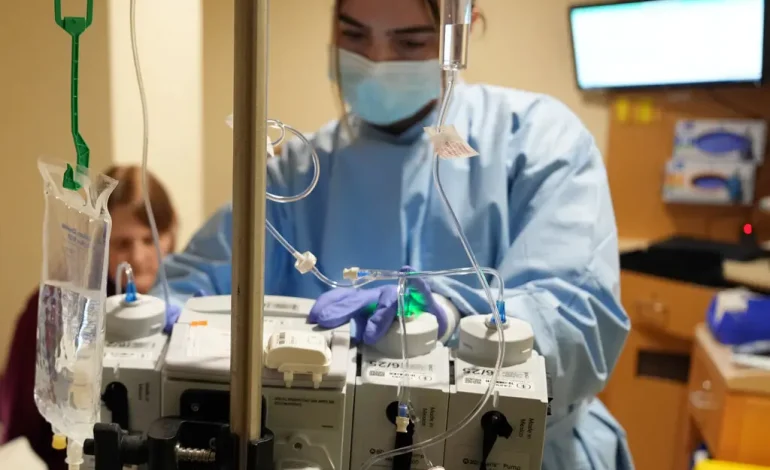
A network of top hospitals and cancer centers that has spent 25 years testing experimental treatments for children with deadly brain tumors is being dismantled after federal funding was cut off — a move doctors say could stall lifesaving research.
The Pediatric Brain Tumor Consortium (PBTC), a coalition of 16 academic centers and children’s hospitals, was told by the National Cancer Institute (NCI) earlier this month that it would not be eligible for renewed funding after March 2026. The NCI provides about $4 million annually, the bulk of the consortium’s budget.
The announcement — delivered not in a formal letter but during a phone call and later confirmed by a brief update on the NCI’s website — left doctors stunned.
“It would seem catastrophic to enroll a child into a trial and then have to say, ‘I’m sorry, you can’t get treatment because the consortium is ending,'” said Dr. Ira Dunkel, a pediatric oncologist at Memorial Sloan Kettering and chair of the PBTC.
Since its launch in 1999, the PBTC has run dozens of early-phase clinical trials for kids with the most aggressive brain cancers — often relapsed patients with grim odds of survival. Unlike late-stage trials that involve hundreds of participants, these trials usually enroll only a few dozen children, testing whether cutting-edge treatments are safe and worth pursuing in larger studies.
Right now, the group has six active trials, including five focused on treatment. One is a groundbreaking CAR T-cell therapy study for recurrent ependymoma — a cancer with no effective treatments. Another, years in the making, was about to begin testing whether laser-based neurosurgery could shrink tumors with less trauma than traditional brain surgery.
“The risk is that we simply won’t do as many impactful clinical trials for pediatric brain tumors,” said Dr. Douglas Hawkins, chair of the Children’s Oncology Group at Seattle Children’s Hospital.
Children already enrolled in PBTC trials will continue treatment, but no new patients can join. Families on waiting lists — often with no other options — are being turned away. Researchers say even if some studies get transferred to another network, such as the Pediatric Early Phase Clinical Trials Network (a broader NCI-backed program for childhood cancers), the transition will cause delays and gaps in access.
That’s a big deal for families facing fast-moving, aggressive cancers.
“We’re talking about patients with very poor outcomes, kids for whom the chance of survival is already very low,” said Dr. John Prensner, a pediatric neuro-oncologist at the University of Michigan.
The NCI has said little publicly, beyond noting on its website that it wants to “optimize resources for maximum impact.” The change comes as the Trump administration continues cutting funds for the National Institutes of Health, though it’s unclear whether the PBTC decision is directly tied to those budget reductions.
The NCI has pledged to ensure current PBTC trials are completed “wherever feasible and appropriate,” but doctors remain doubtful.
“Some studies may close prematurely,” Dr. Dunkel admitted.
Beyond the loss of active studies, physicians worry about something harder to measure: the collaboration. The PBTC created a rare hub where pediatric oncologists, neurosurgeons, and scientists swapped ideas and built research together.
“The intangible loss is the cross-fertilization — the brainstorming, the collaboration, the sparks that happen in these groups,” said Dr. Mark Souweidane, director of pediatric neurosurgery at Weill Cornell and Memorial Sloan Kettering.
For families navigating a child’s brain cancer diagnosis, those sparks often led to the only ray of hope. Without them, experts fear progress will slow — and the kids who need new treatments most will have fewer chances to find them.
The original story by Nina Agrawal for the New York Times.







The latest news in your social feeds
Subscribe to our social media platforms to stay tuned